By Ikugbadi Oluwasegun
The National Agency for Food and Drugs Administration and Control (NAFDAC) has notified healthcare professionals and the public of a confirmed counterfeit batch of Mabthera 500mg/50ml cancer drug circulating in Nigeria.
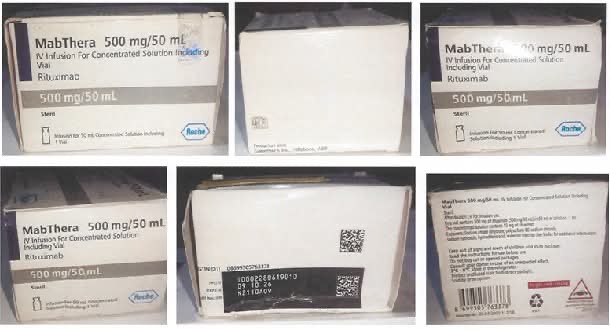
This alert follows official communications from Roche Nigeria confirming counterfeit cases of Mabthera 500mg/50ml with batch number N2110A09 reported in Kaduna and Gombe states of Nigeria.
According to the Marketing Authorisation Holder (MAH), the report of a counterfeit batch of Mabthera 500mg/50ml was received following complaints from healthcare professionals in both locations mentioned above.
In both cases, the counterfeit batch of the product was brought in by patients at each location and reportedly sold at significantly lower prices, between NGN 160,000 and NGN 275,000, respectively.
Roche Nigeria’s investigation revealed clear evidence of counterfeit activities regarding the folding box. The following differences were detected:
- The batch number does not exist in the Roche database.
- Wrong language (Turkish instead of English)
- Wrong placement of Braille text
- Incorrect tamper-evidence labels.
In addition, several significant differences were detected on the labelled vial, including:
- A non-existent batch number
- Wrong language (English instead of Turkish)
- Different glass vial (e.g. shape and dimensions)
- Different stopper (e.g. colour, dimensions)
- Different aluminium seal (e.g. dimensions)
- Different flip-off cap (e.g. colour, shape)
Chemical analyses were not possible as investigations were conducted based on packaging photographs sent in by complainants; no physical samples were available for return to Roche.
Mabthera (rituximab) is an antibody injection used to treat blood cancers (non-Hodgkin’s lymphoma, chronic lymphocytic leukaemia), severe rheumatoid arthritis, and autoimmune diseases like pemphigus vulgaris and vasculitis. It destroys specific white blood cells (B-cells) that cause disease.
Note: The correct manufacturing site for genuine Mabthera 500 mg/50 mL is Made for F. Hoffmann-La Roche Ltd, Basel, Switzerland, by Roche Diagnostics GmbH, Galenical Plant, Germany, Sandhofer Strasse 116, Mannheim 68305.


























