By Ikugbadi Oluwasegun
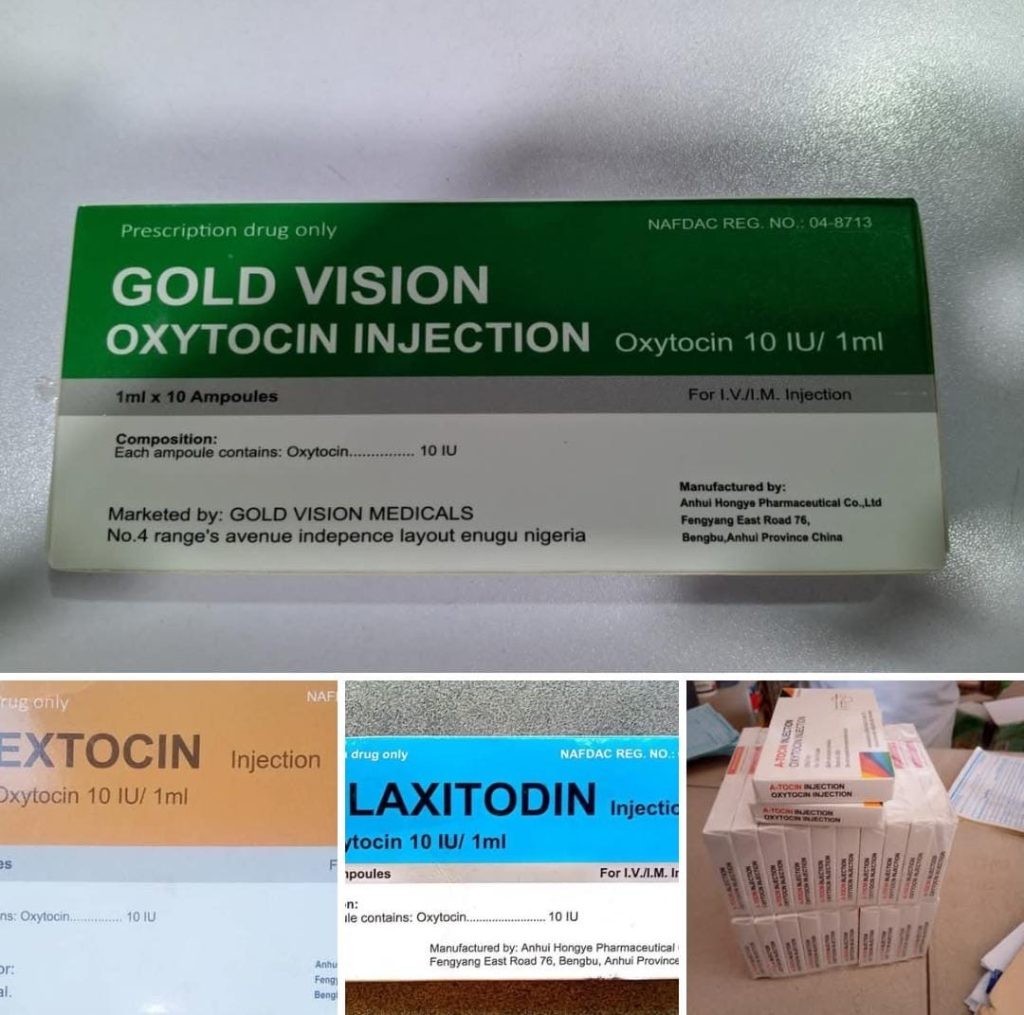
NAFDAC hss alerted the public of an unregistered Gold Vision Oxytocin injection 10IU with a fake NAFDAC Registration Number A4-9566. The product is purportedly manufactured by Anhui Hongye Pharmaceutical Co., Ltd., located at Fengyang East Road, Bengbu, Anhui Province, China, and marketed by Gold Vision Medicals, No.4 Range Avenue, Independence Layout, Enugu, Nigeria.
The product was discovered during a Risk-Based sampling survey activity conducted by officers of NAFDAC.
Further investigation revealed that three (3) falsified products, namely A-tocin injection, Extocin Injection, and Claxitodin injection, were discovered during the 2023 RBPMS sampling.
The products were reportedly manufactured by the same company, and all the products bear the same falsified NAFDAC Registration Number A4-9566.
It is essential to note that the products identified in this alert are confirmed as falsified because they are not listed in the NAFDAC-registered products database.
Risk Statement
Using unregistered or falsified Oxytocin injections poses serious health risks to both mothers and newborns.
These products may contain incorrect doses, no active ingredient, or harmful contaminants, leading to ineffective uterine contractions, postpartum haemorrhage (PPH), and even maternal death. Poor-quality oxytocin may also delay or fail to stop bleeding after childbirth, increasing the need for emergency interventions like blood transfusions or surgery.
Healthcare professionals and consumers are advised to report any suspicion of the sale of substandard and falsified medicines or medical devices to the nearest NAFDAC office, call 0800-162-3322, or send an email to sf.alert@nafdac.gov.ng.